In women undergoing primary cesarean section, the administration of alpha lipoic acid improves uterine healing and decreases the incidence of scar niches.
The aim of this parallel-group, triple-blind, placebo-controlled randomized clinical trial was to investigate the effect of alpha lipoic acid (ALA) on uterine wound healing after primary cesarean section (CS).
To do so, 102 women undergoing pre-labor primary CS were randomized to receive either oral ALA (Thiotacid® film-coated tablet 600 mg, twice daily) or a placebo twice daily for 6 weeks after CS.
Results showed that twenty (39.2%) women in the treatment group and ten (19.6%) controls had no niche (p = 0.03). The absolute and relative risk reduction of forming a niche was 19.61% and 24.39%, respectively. The number of women needed to treat was five to avoid one niche formation. ALA use was associated with gastrointestinal upset in only three participants; however, none of the women withdrew during the study.
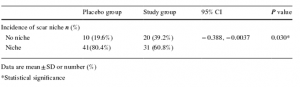
The cesarean section scar-related measurements in the two groups.
Authors concluded that women undergoing primary CS, the administration of ALA for 6 weeks postpartum improved uterine healing and decreased the incidence of scar niche.
Sammour H, Elkholy A, Rasheedy R, Fadel E. The effect of alpha lipoic acid on uterine wound healing after primary cesarean section: a triple-blind placebo-controlled parallel-group randomized clinical trial. Arch Gynecol Obstet. 2018 Dec 18.
